Automation of Dissolution Tests
for Biowaiver Studies
Rolf Rolli
SOTAX Group, Allschwil, Switzerland
email for correspondence: rolf.rolli@sotax.ch
Recent research has lead to the use of in-vitro tests to waive
additional in vivo bioequivalency studies for some pharmaceutical
products. The use of in vitro testing to achieve a waiver of in
vivo studies is commonly referred to as a biowaiver.This article
describes the use of automated dissolution instrumentation and
software for biowaiver studies.
Introduction
Speed of drug introduction to the marketplace is a key success
factor for a pharmaceutical company. Fast development and launch
of a drug result in significant economic gains for a drug manufacturer.
A recent FDA guidance based on a Biopharmaceutics Classification
System (BCS) permits waiver of in vivo Bioavailability and Bioequivalencey
studies. The Biopharmaceutics
Classification System classifies drugs as follows:
Class 1: High Solubility - High Permeability
Class 2: Low Solubility - High Permeability
Class 3: High Solubility - Low Permeability
Class 4: Low Solubility - Low Permeability
In vitro tests for solubility, permeability and dissolution
form the basis of a drug product's classification and qualification
for biowaivers.
Current regulations permit waivers for Class 1 rapidly dissolving
drugs, which represent up to 25% of the drugs. Efforts in science
and regulatory bodies are being made to extend biowaivers to certain
Class 2 and 3 products, which would represent more than 50% of
all drugs coming on the market.
In vivo bioavailability and bioequivalencey studies cost up
to $ 250,000 each and can require up to 2 months to complete.
On the other hand, in vitro laboratory tests are rather inexpensive
and fast. Dissolution studies, f 2 profile calculations and report
writing represent a large part of the laboratory work. SOTAX offers
an automated dissolution system with software designed for biowaiver
testing. The fully automated Dissolution test system AT 70smart
can perform multiple runs with different media/pH. (Figure 1)

Figure 1: SOTAX AT 70smart
With this system up to 10 USP 2 tests can be fully automated,
from the tablet input up to the print out of the report.
The AT 70smart is designed to:
· Dispense 3 different media and water
· deliver the medium to all vessels simultaneously
· drain and clean all vessels simultaneously
· deaerate medium with helium sparging
· remove sample aliquots using the hollow shaft sampling
system
· allow the use of different filter membranes
· analyze samples using on-line UV or off-line HPLC
· perform up to10 tests.
Test with baskets require the Basket-Station BS 60. (Figure
2) Up to 10 USP 1 tests can be loaded.

Figure 2: SOTAX AT 70smart with Basket station BS 60
The system is thoroughly cleaned after each test using water.
Prior to initiating the next test the vessels are pre-rinsed
with the selected test medium. The programmed cleaning procedure:
· drains the medium from the vessels through valves at
the bottom of the vessels
· rinses the vessels multiple times with rotating spray
heads at 4 bar pressure allowing the removal of any sticky residue
· cleans all tubing and filter stations using high pressure
reversed sparging
· removes any remaining liquid in lines with a final air
purge.
This cleaning procedure significantly reduces the risk of carry
over and cross contamination.
The AT70smart system utilizes CFR 21 Part 11 compliant software,
WinSOTAX. WinSOTAX software for biowaiver testing contains standard
check boxes for test type, medium type and volume selection, speed
and sampling time. Check boxes are activated and used for each
parameter setting. The regulations require the testing of 12 test
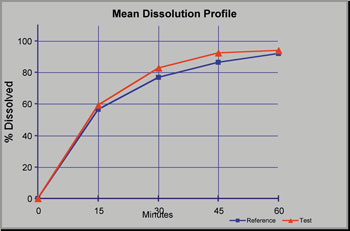
samples and 12 references samples for dissolution profile comparison.
Similarity factor, f2 calculations, are performed on the stored
results and a report is generated with comparison profiles, f2
calculations, statistics and a pass/fail comment for the dissolution
profile similiarity (f2) criteria.
The WinSOTAX software offers the following user input options:
Method Generator Check boxes:
__USP Apparatus I at 100 rpm (Default)
__USP Apparatus II at 50 rpm (Default)
__Immediate Release Product
__Extended Release Product
Selectable media:
__0.1 N HCl, 900 ml (Default)
__pH 4.5 buffer, 900 ml (Default)
__pH 6.8 buffer, 900 ml (Default)
Any other media may be added
Pre-programmed sampling points:
__Immediate Release products 10, 15, 20, 30 and 60 minutes
__Extended Release products 1, 2, 3, 5 and 8 hours