LOGAN INSTRUMENTS Launched THE PERMETRO SYSTEMS in International Markets
A Dissolution and Permeation All-In-One System
The Logan PEMETRO system uses a bionic intestinal membrane to simulate the release and absorption of drugs in the gastrointestinal tract in human body, providing a dynamic absorption profile for permeation through dissolution cycle, and vividly mimicking the in-vivo drug absorption in intestinal for the first time. The PERMETRO is fully automated and works seamlessly with all USP dissolution apparatus, e.g., 1, 2, 3, or 4.
Logan PERMETRO 1200 is designed for simultaneous dissolution or permeation tests; special program is included to study the bioequivalence (BE). Its features include: to check drugs’ permeability; to compare permeability between generic and reference listed drug (RLD) permeability; and to check dissolution rate and permeation rate at the same time.
Logan PERMETRO 3700 is specially designed to study the drug permeation under different buffer pH in addition to the parallel dissolution / permeation tests. Features include: compare control release products permeability, check permeability between generic and RLD, and check dissolution and permeation rates at the same time.

Ever since LOGAN proudly announced that PERMETRO - “A dissolution and permeation all-in-one automated testing system” was available globally for all customers at AAPS 2019 in San Antonio USA, they have been successfully operated in a few labs. One laboratory in India is currently using LOGAN PERMETRO to test the drug permeation before the bioequivalence (BE), the other pharmaceutical companies in China are using it to compare their generic products against the RLD. The test data are shown below:
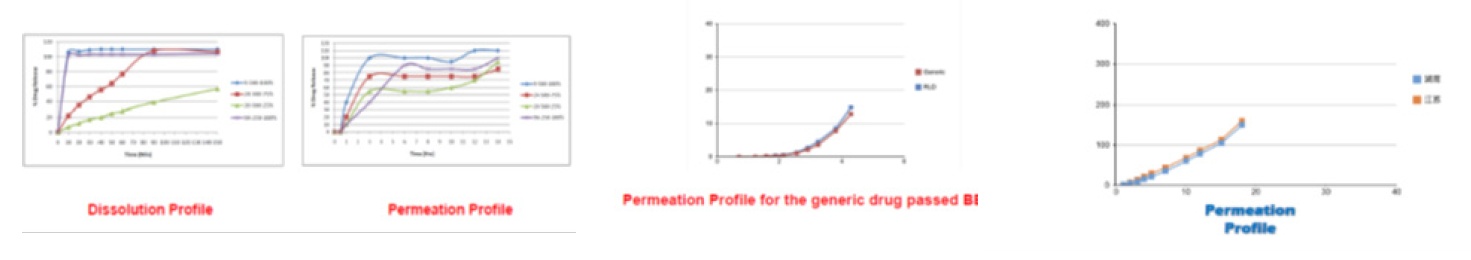
Logan’s PERMETRO system is the most advanced in the pharmaceutical industry, introducing a new way for drug development, including in vitro in vivo correlation (IVIVC), BE, and the interferences of food or drugs on drug absorption. Logan is also accepting samples for dissolution/permeation tests. For more information, please contact Ms. Joan Lee (TEL: 732 302 9888 ex 105) at Logan Instruments Corp.
