Questions and Answers November 2021
Margareth R. Marques, Ph.D. and Mark Liddell, Ph.D.
The following questions have been submitted by readers of Dissolution Technologies. Margareth R. Marques, Ph.D., and Mark Liddell, Ph.D., United
States Pharmacopeia (USP), authored responses to each of the questions. *Note: These are opinions and interpretations of the authors and are not
necessarily the official viewpoints of the USP.
Email for correspondence:
mrm@usp.org
Q In the USP general chapter <724> Drug Release, under USP Apparatus 5, when using a watch glass (90 mm) with a polytef screen (sandwich), what is the best way of determining the paddle height? The chapter text states that “the disk holds the system flat and parallel to the bottom of the paddle, with the release side up.”. Figure 1a from the chapter shows how to setup the paddle height but using a flat screen. A polytef screen normally follows the contour of the glass and sample and is not parallel to the paddle.
Figure 1a from USP general chapter <724> Drug Release: 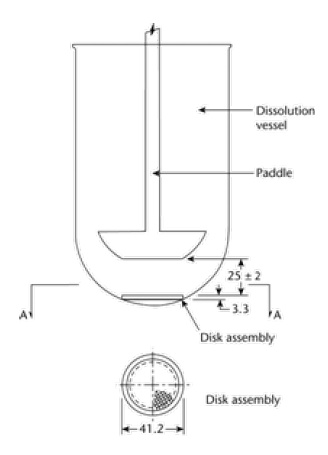
We have concluded there are 3 possible outcomes shown in the figure below.
In the left figure, the paddle is set at 25 mm above the rim of the watch glass/outer diameter of the polytef screen. This is a parallel surface. In the middle figure, the sandwich is a spherical setup and the height changes per sample used. The height is measured when the sample is in place and on top of the screen. This is difficult and calibration of the height can contaminate the sample. In the drawing on the right, using the watch glass with polytef screen and setup the height is 25 mm above the bottom of the screen. The setups shown in the figures on the left and right can be done without disturbing any sample and can be easily done with the use of a calibration tool. 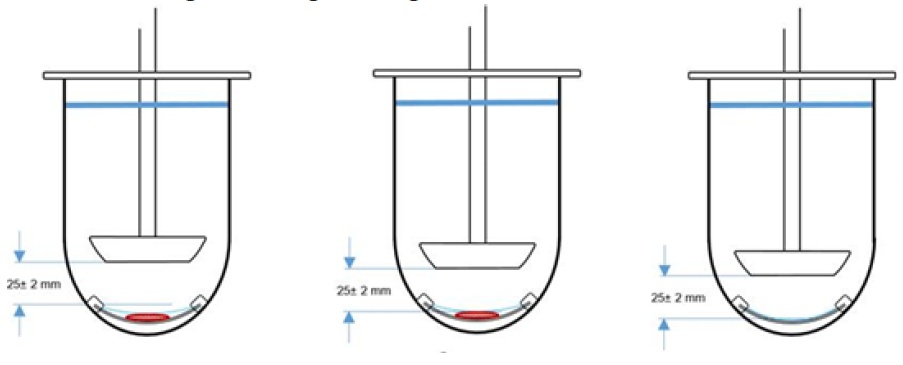
A Without a full characterization of the system and procedures it would be difficult to determine a priori which paddle heigh measurement would provide the most reproducible results; however, a basic principle of dissolution / drug release testing is that the test system and parameters selected should provide robust and reproducible test conditions. With that in mind, considering the Figure 1a in <724>, the 2.5 cm is measured from the flat disk at the center of the assembly. Using the drawings provided, the most reproducible approach to setting the height of the paddle is to use the approach described in the drawing on the right, where the measurement is made from the center of the assembly.
Q We manufacture suspensions, and we use a composite sample to perform the dissolution. The composite sample is made by mixing a (n) unit dose representing beginning, middle, and end, and most of the unit doses we make are 5 mL. Is this procedure appropriate?
A According to the USP general chapter <1711> Oral Dosage Forms - Performance Tests and the FDA productspecific guidances, the suspension should be prepared/mixed according to the instructions given to the patient, and a sample equivalent to the highest unit dose administered at one time should be transferred to each vessel/unit of the dissolution equipment.
Q We evaluated the revised USP monograph for Oxcarbazepine Oral suspension and noticed that dissolution test 2 is newly proposed. T he dissolution test 2 directs to use a separate bottle of oral suspension for each vessel. We would like to know the rational for such procedure.
A The rationale for use of independent bottles for testing dissolution of suspensions is described in the USP general chapter <1711> Oral Drug products - Performance Tests and in several FDA product-specific guidances.
Q As per USP chapter <711> Dissolution, under Apparatus 1 and 2, it is mentioned: “The distance between the inside bottom of the vessel and the bottom of the basket/blade is maintained at 25 ± 2 mm during the test.” As the height of different volume vessels are different, how should we adjust this height of 25 ± 2 mm during the test?
A There are several tools available that can be used to set up the equipment and check the vessel and paddle alignment and height. The figure below shows a cut away vessel with an example of a height gauge that can be used to determine the stirring element height.

Check with the supplier of your dissolution equipment. They may have this height gauge or a similar measuring device available for purchase. You can also refer to the website https://www.usp.org/pvt/dissolution-instrument-qualification for a description of best practices associated with the setup and Performance Verification Test (PVT) of the USP basket and paddle dissolution apparatus.
Q Can you explain the reason for including “degassed with helium” under the dissolution medium for USP monograph of Cabergoline Tablets? Secondly, if we use simple 0.1N HCl without degassing, then what will be the impact, because the British Pharmacopoeia monograph does not state to degas the medium with helium.
A Here is a screen shot from the USP Carbegoline Tablets monograph:
The cross reference to Dissolution <711> indicates that the general procedure from the general chapter <711> should be followed. All parameters that are product-specific are defined in the monograph text.
The dissolution, disintegration, and drug release tests in the USP - NF are the tests approved by the United States FDA for the product to be marketed in the USA. Although it is possible to deaerate the dissolution medium using and alternative procedure (there are several methods available for degassing dissolution media), whatever procedure is used should be demonstrated to be effective when compared to the reference procedure. For example, there is an alternative deaeration procedure described in general chapter <711> Dissolution and other procedures have been described and evaluated in Dissolution Technologies (www.dissolutiontech.com).
One should keep in mind that degassing or deaeration of dissolution media is not mandatory for all dissolution tests. Degassing or deaeration of the dissolution medium should be evaluated on a case-by-case basis. (See USP general chapter <1092> The Dissolution Procedure: Development and Validation). It is entirely possible that the degassing effect may be specific to the product or formulation.